  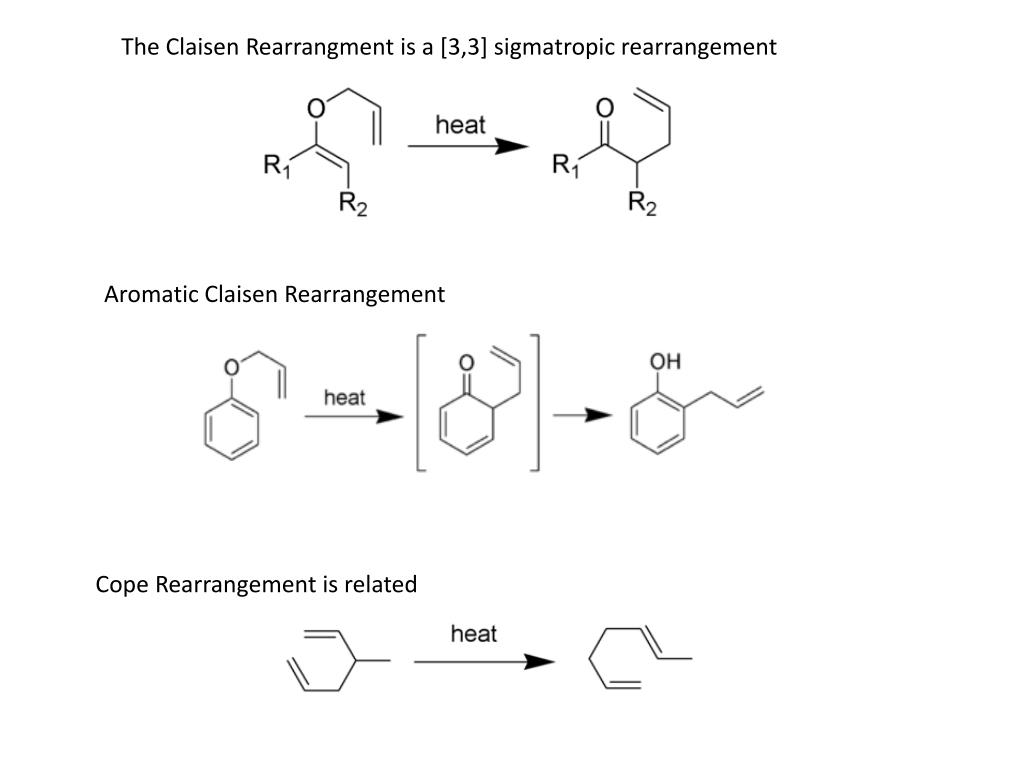
A chapter which summarises the bedside rules and equations used in the interpretation of blood gases is also available as a brief overview of the empirically derived formulae which describe acute and chronic compensation for acidosis and alkalosis. The physiological consequences of acidaemia and alkalaemia are discussed in dedicated chapters, as are the various effects of having an excessively high or precipitously low blood CO 2 level (independent of pH changes). The considerable acid strength of intact carbonic acid indicates that it is an important protonation agent under physiological conditions.This chapter focuses on the ways in which a changing CO 2 concentration might alter the pH of a solution, particularly that of your precious bodily fluids. This establishes H2CO3 as being 0.37 pKa units stronger and about 1 pKa unit weaker, respectively, than the physiologically important lactic and pyruvic acids. 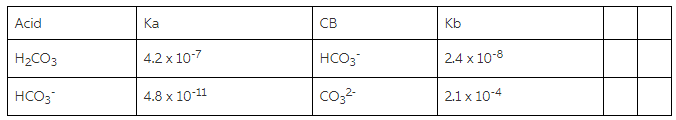
The pKa of carbonic acid was found to be 3.49 ± 0.05 using both the Marcus and Kiefer-Hynes free energy correlations. The on-contact PT rates were found to follow the acidity order of the carboxylic acids: the stronger was the acid, the slower was the PT reaction to its conjugate base. The time-dependent diffusion-assisted PT rate was analyzed using the Szabo-Collins-Kimball equation with a radiation boundary condition. The on-contact proton transfer (PT) reaction rate from the optically excited photoacid to the carboxylic bases was derived, with unprecedented precision, from time-correlated single-photon-counting measurements of the fluorescence lifetime of the photoacid in the presence of the proton acceptors. A particular goal is to establish the pKa of carbonic acid H2CO3. Carbonic, lactic, and pyruvic acids have been generated in aqueous solution by the transient protonation of their corresponding conjugate bases by a tailor-made photoacid, the 6-hydroxy-1-sulfonate pyrene sodium salt molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
